Welcome to BasalFix
Who We Are
At BasalFix, we pride ourselves on offering a state-of-the-art dental implant system that is entirely developed and manufactured in Italy. Our commitment to quality is reflected in our use of certified medical materials sourced from reputable suppliers.
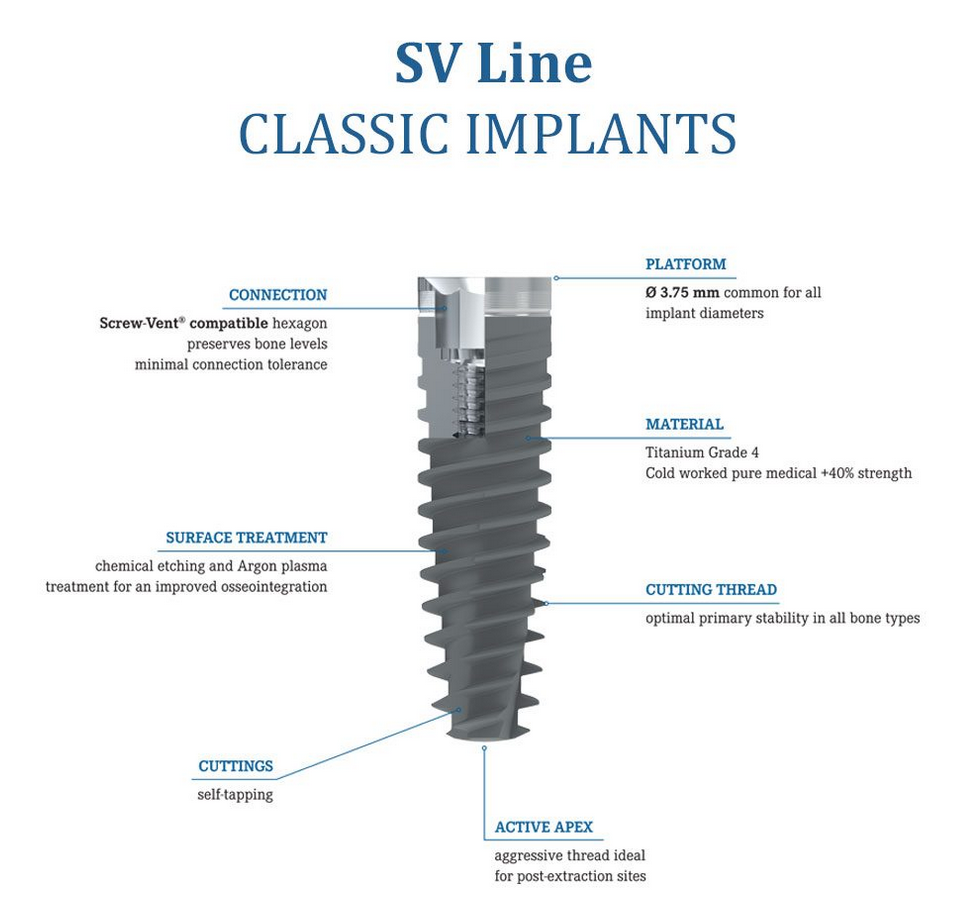
BasalFix is a premier dental implant system, entirely developed and manufactured in Italy, utilizing only certified medical materials sourced from reputable suppliers. Each BasalFix implant is crafted from high-strength Titanium Grade 4, in compliance with ASTM F 67 standards.
Our Implants
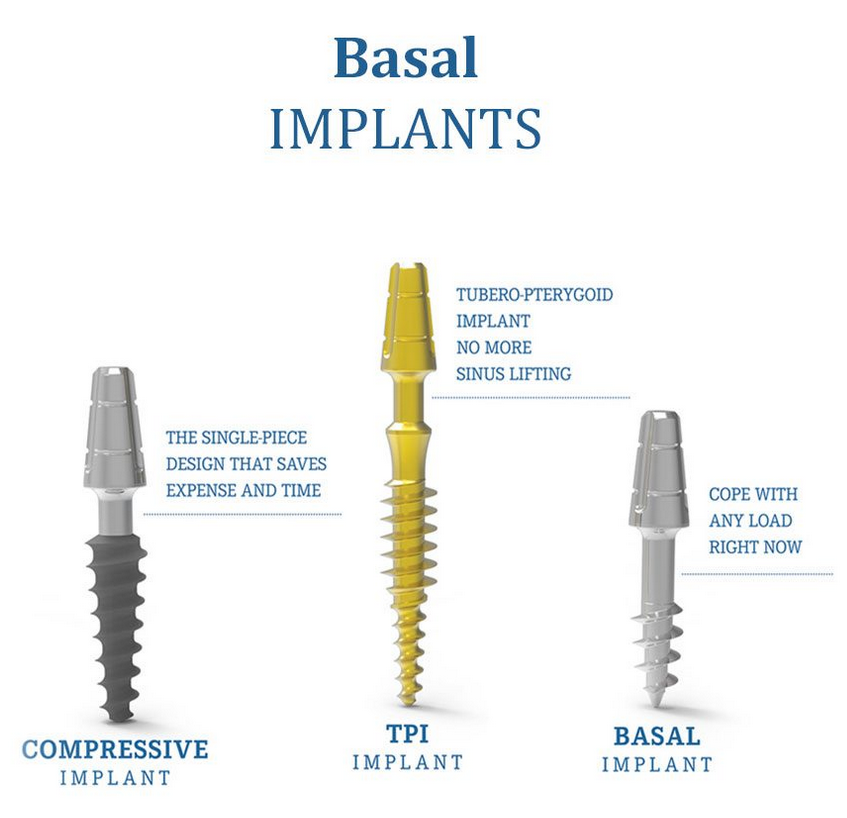
BasalFix Implants are crafted from high-strength titanium grade 4, adhering to the ASTM F 67 standards, and produced in an ISO 7 certified clean-room environment. This ensures both the safety and durability of our implants. Each BasalFix dental implant is a single-piece construction available in various sizes, featuring the most flexible abutments for a perfect fit. They are indicated for multiple rehabilitation only, suitable for both flap and flapless techniques.
Quality and Assurance
We are dedicated to providing the best price-quality ratio in the market. With every BasalFix implant, you receive a passport that guarantees authenticity and traceability, ensuring peace of mind for both practitioners and patients.
Compliance
Basalfix medical devices comply with the European Regulation
MDR 2017/745. The manufacturer Biomec S.r.l. has been equipped
with a quality system ISO 9001 and ISO 13485 since 1998.
Experience the excellence of BasalFix, where innovation meets reliability in dental implant technology.
Comprehensive Solutions:
Types of Implants: Basal, TPI, Compressive, Classic
Prosthetic Components: High-quality parts for perfect dental restorations
Surgical Instruments: Precision tools for optimal surgical outcomes